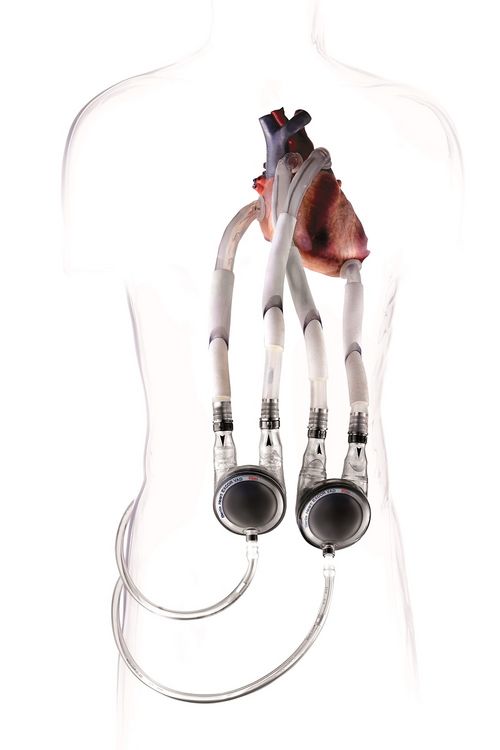
The EXCOR® Adult system comprises components that optimally match one another and have proven successful in practice:
Silicone Cannulae
Our EXCOR® cannulae are made by hand from the purest medical silicone. In order to optimally supply patients, the cannula range includes cannulae of various types and sizes with the following features:
- Highest biocompatibility
- Customization to diverse anatomical Customization to diverse anatomical conditions thanks to various diameters, lengths, angles and tip styles
- Arterial and/or atrial cannulae with flexible metal reinforcement for adaptation to the individual anatomy
- Velour-coated suture ring, developed for a fast and safe anastomosis
- Good ingrowth of the cannula to the transcutaneous exit site thanks to the velour sheathing
Blood pumps
The EXCOR® Adult range includes blood pumps of various sizes, which are optimally suited to patients’ needs. Our blood pumps boast the following features:
- Different sizes available: 50, 60 and 80 ml pump volumes
- Transparent polyurethane casing allows for visual inspection: pump filling and any deposits can be detected
- Flexible triple-layer membrane for a high degree of safety; graphite powder between the membrane layers minimizes friction
- Ultra-smooth, flow-optimized blood contact surfaces with Carmeda® BioActive Surface* coating for the best possible blood compatibility
- Safe de-airing through a de-airing port
- Valves at the inflow and outflow section guarantee that the blood flows in the right direction
- Selection of blood pumps with tri-leaflet valve made from polyurethane or blood pumps with bileaflet valve made of carbon
Stationary driving unit Ikus
The Ikus driving unit is designed specifically for stationary use with EXCOR® blood pumps. As a high-performing all-rounder, the Ikus covers all areas of application:
- Pump rate, operating mode, driving pressure variable within wide limits
- Biventricular operation in synchronous mode, asynchronous mode or with independent control of the pumps
- Laptop with permanently installed monitor program for system configuration and monitoring
- High degree of safety due to multiple redundancy
- Automatic 30-minute battery power supply in the event of mains power failure
- Patient transport within the hospital while in battery operation
- Acoustic and visual alarms
- Plain text messages in the monitor program
- Comprehensive system status information at a glance
Mobile Driving System Excor
The mobilization of the patient is important for his/her well-being and the success of the therapy. With the Excor mobile, mobilized patients can move freely and can usually be discharged home. The mobile driving unit offers a high degree of safety and user-friendly handling:
- Can be used for 60 ml and 80 ml EXCOR® blood pumps
- Optimal operating point within pre-set limits
- Synchronous pump with simultaneous pressure build-up comparable to native heart contractions
- Lightweight
- Acoustic and visual alarms in the event of faults
- Two rechargeable batteries and two additional backup rechargeable batteries for several hours of mains-free operation
- Clear display on each battery: charge level is displayed
- Emergency batteries in the drives for additional safety
- Modular construction allows the patient to replace components