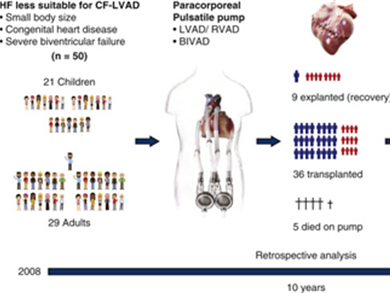
A study by the Swedish Sahlgrenska University Hospital in Gothenburg shows the positive outcomes of the EXCOR® therapy:
“Considering the severity of patient illness and disease complexity in many of our cases, the use of the paracorporal EXCOR as a bridge to transplantation or recovery resulted in a high survival. In addition, the overall long-term survival after HTx or recovery was remarkably good.“
According to Bartfay et al. the EXCOR® VAD is a viable strategy for children with a small body size, patients with complex congenital heart diseases and patients requiring biventricular heart support (BVAD). „Due to its versatility as a circulatory support system our findings suggest that the EXCOR® pump is a valuable option in patients ineligible for CF-LVAD.“
EXCOR is the only system approved for pediatric and BVAD use.
Read more about: www.sciencedirect.com/science/article/abs/pii/S0022522320311661
The access to some or all shown products may be restricted by country-specific regulatory approvals. The use of EXCOR® VAD for adults, RVAD-support, Excor mobile and EXCOR® Active is not FDA-approved.